|
Then, by selecting the HOMO/LUMO option in the Compare mode, the database will display the HOMO of the currently selected molecule with the LUMO of the molecule chosen from the molecule comparison list. Reaction Predict from the Optional Views menu. In order to compare the HOMO of one molecule with the LUMO of another, select M.O. If two constructive interactions are possible, the one with the lower energy difference between the HOMO and LUMO is more likely to occur. Constructive overlap is possible between the blue region of an occupied orbital and the yellow region of an unoccupied orbital or between the green region of an occupied and the red region of an unoccupied orbital. In this program, color codes for the wave function signs are arbitrary. In the regions of large overlap the orbitals (wave functions) must have the same sign. 
In order for bonds to form, the overlap of the orbitals must be constructive. According to the definition of HOMO/LUMO by the International Union of Pure and Applied Chemistry (IUPAC), HOMO is the Highest-energy Occupied Molecular Orbital (filled or partly. It is often through overlap of the HOMO of one molecule with the LUMO of another that new bonds are formed during chemical reactions. In this report, we synthesized differently substituted 1,3,5-triazine based organic molecule which on future processing can be used in. The 3-D calculated molecular orbitals are shown in Figure 10.5.6. >you should always use orbitals and orbital energies obtained at optimized geometry. 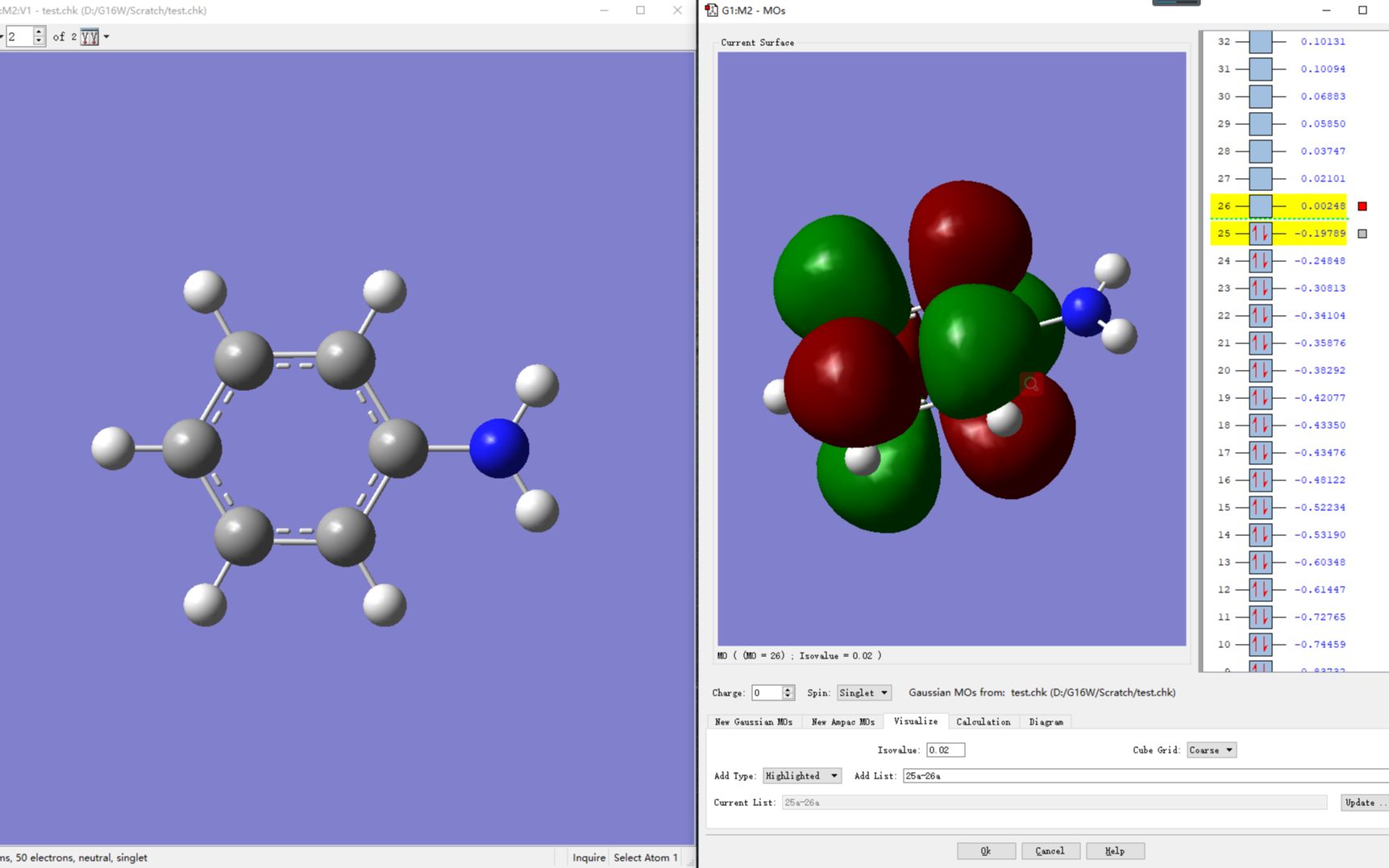
The energy difference between the HOMO and LUMO is termed the HOMOLUMO gap. Vacuum level Ca (2.8 eV) 3 Desirable LUMO energy range Al (4.1 eV) of LUMO level Au (5 eV) Eg 5 Desirable 4 HOMO energy range 6(a) Pt (6 eV) of HOMO. All molecules have a HOMO (highest occupied molecular orbital) and a LUMO (lowest unoccupied molecular orbital). HOMO and LUMO are acronyms for highest occupied molecular orbital and lowest unoccupied molecular orbital, respectively and are often referred to as frontier orbitals.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
